
Solta Medical Clinical Insights
Peer-reviewed evidence, treatment protocols, and clinical education resources. We publish the data — you decide whether our platforms meet your clinical standards.

Peer-reviewed evidence, treatment protocols, and clinical education resources. We publish the data — you decide whether our platforms meet your clinical standards.
Solta Medical platforms are supported by one of the largest bodies of peer-reviewed evidence in the aesthetic energy device category. Below is a transparent summary of what the literature shows — including where evidence is strong and where further study is ongoing.
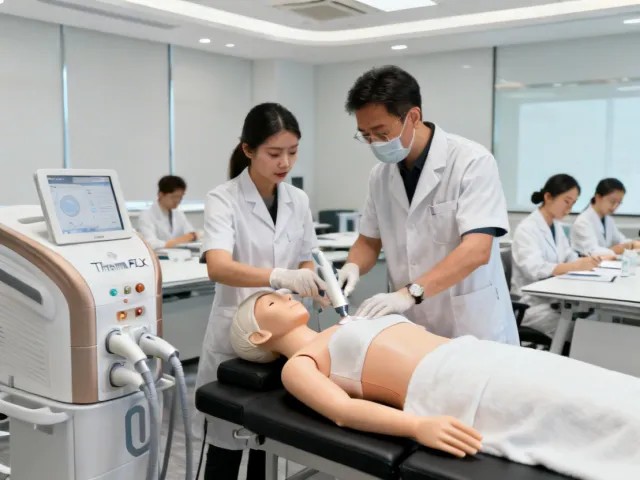
Every Solta Medical platform purchase includes a comprehensive clinical training program. We believe that device outcomes depend as much on operator technique as on the technology itself — so we invest heavily in education.
Two-day hands-on program at your facility. Covers device operation, treatment planning, patient selection criteria, and complication management. Conducted by a certified Solta Medical clinical education specialist with a minimum of 5 years of aesthetic practice experience.
Five dedicated training facilities across the United States (Hayward CA, Dallas TX, New York NY, Chicago IL, Miami FL). Multi-day advanced courses cover complex cases, combination protocols, and practice integration strategies.
24/7 access to treatment protocols, video case libraries, parameter guides, and live webinar series. Updated quarterly with the latest clinical evidence and technique refinements. Available to all active Solta Medical device owners at no additional cost.
Regional peer study groups and an annual Solta Medical Clinical Summit where practitioners share outcomes, discuss challenging cases, and learn from high-volume users across specialties.
A representative sample from our clinical evidence library. Full references available upon request.
Journal of the American Academy of Dermatology, 2019; 81(4): 1034-1042
150-subject RCT demonstrating statistically significant improvement in skin laxity at 6-month follow-up with fourth-generation monopolar RF platform.
Dermatologic Surgery, 2018; 44(11): 1457-1468
Meta-analysis of 12 studies (n=456) confirming 51-75% improvement in atrophic scarring with non-ablative fractional laser across 3-5 treatment sessions.
Aesthetic Surgery Journal, 2017; 37(suppl_3): S114-S121
Prospective comparison showing 53% less post-operative bruising and 36% faster recovery with ultrasonic-assisted technique versus suction-assisted liposuction.
Lasers in Surgery and Medicine, 2021; 53(8): 1087-1094
Retrospective analysis (n=200) demonstrating sustained improvement at 24 months in 70% of subjects, with gradual return to baseline by 36-48 months.
Request access to our complete clinical evidence portfolio, including white papers, treatment parameter guides, and case study documentation.
Request Clinical Materials